Vanguard 5L4 Plus is a vaccine that is intended for dogs to avoid distemper, parvovirus eneritis, parvovirus, infectious hepatitis of frugivores, leptospirosis, respiratory diseases, parainfluenza, coronavirus. The vaccine is used for dogs starting from the 6th week and older. Many veterinarians advise using these vaccines to vaccinate dogs, because they are well absorbed by animals, and the immunity developed with the help of the vaccine will be in excellent condition for at least a year.
Specifics of the Vanguard vaccine
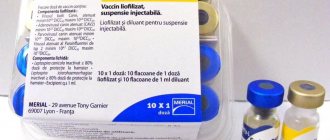
One dose of the vaccine is obtained by mixing the components from two glass vials containing substances of different consistency, with volumes equal to one cubic centimeter.
Let's take a closer look:
The dry component is a lyophilized mass of white, beige or light yellow shades, which is characterized by a porous structure. The substances that make up its basis are weakened strains that cause plague, enteritis of various etiologies, and parainfluenza in animals. Auxiliary components of the contents of the bottle are substances that preserve viruses. These include: solutions of gelatin and sucrose, gentamicin, Hanks' medium. The liquid component is basically aluminum hydroxide and has a transparent or light pink color. The bottle with the liquid contents of the Avant-Garde vaccine contains inactivated pathogens of leptospirosis and is used to dissolve the dry lyophilic mass.
The ampoules of the Vanguard system are hermetically sealed with rubber stoppers, on top of which aluminum caps are rolled. Supplied in plastic containers, each containing 25 doses (50 vials in total). In pharmacy chains, the vaccine for dogs with coronavirus is sold individually.
Operating principle of Vanguard:
Vanguard is an American vaccine developed by , the components of which ensure the production of antibodies within 21 days after administration of the dose. Immunity formed through vaccination provides reliable protection against parainfluenza and canine distemper, infectious hepatitis, parvovirus enteritis, leptospirosis, adenoviral and coronavirus infections. Vanguard's activity persists for 12 months, after which additional preventive vaccination will be required, which should be repeated annually.
Vaccine Vanguard Plus 5 L4 CV, dose, vial. 1ml
COMPOSITION AND RELEASE FORM The vaccine consists of two separately packaged components: The dry component is made from attenuated causative agents of canine distemper (strain N-CDV), canine adenovirus infection (serotype II Manhattan strain), carnivorous parainfluenza (strain NL-CP1-5), parvovirus enteritis dogs (strain NL-35-D), thiomersal-inactivated leptospira serogroups Canicola (strain C51), Grippotyphosa (strain MAL 1540), Icterohaemorrhagiae (strain NADL 11403) and Pomona (strain T262) with the addition of gentamicin as a preservative (up to 30 μg/ ml), stabilizers (NZ Amine AS (160 g/ml), gelatin solution, sucrose solution), antifoam and Hanks' medium. The liquid component of the vaccine is made from the causative agent of canine coronavirus (strain NL-18), inactivated with thiomersal with the addition of aluminum hydroxide and Hanks' medium as an adjuvant. In appearance, the dry component is a homogeneous porous mass of yellow-white color, the liquid component is a homogeneous suspension of light pink color. The vaccine components are packaged in 1 cm3 (one dose) in sterile glass or plastic vials, hermetically sealed with rubber stoppers reinforced with aluminum caps. Vials with vaccine components are packaged in plastic boxes of 25 doses (50 bottles). Each box of vaccine contains instructions for use. BIOLOGICAL PROPERTIES The vaccine causes the formation of an immune response in dogs to the causative agents of plague, infectious hepatitis, parainfluenza of carnivores, adenovirus and coronavirus infections, parvovirus enteritis and leptospirosis of dogs 21 days after repeated administration, lasting at least 12 months. One immunizing dose of the dry component contains at least: 102.5 TCID50 of canine distemper virus, 102.9 TCID50 of adenovirus type II, 105.0 TCID50 of canine parainfluenza virus, 107.0 TCID50 of canine parvovirus, 600 NU Leptospira canicola, 600 NU Leptospira grippotyphosa , 600 NU Leptospira icterohaemorrhagiae, 600 NU Leptospira Pomona. One immunizing dose of the liquid component contains at least 1.49 RP of canine coronavirus. The vaccine is harmless and has no medicinal properties. INDICATIONS Prescribed to puppies and adult dogs for the prevention of distemper, infectious hepatitis, adenovirus and coronavirus infections, parainfluenza of carnivores, parvovirus enteritis and leptospirosis. DOSES AND METHOD OF ADMINISTRATION Healthy dogs are subject to vaccination starting from 6 weeks of age. The vaccine is administered subcutaneously three times with an interval of 3 weeks in a volume of 1.0 cm3 (one immunizing dose). For primary immunization of animals at the age of 9 weeks, two-time vaccination is allowed with an interval of 3 weeks. Dogs are revaccinated once a year with one dose of vaccine. Adult dogs that have not previously been immunized are vaccinated twice with an interval of 2-3 weeks. Before use, the contents of the bottle with the liquid component are shaken, added to the bottle with the lyophilized component and mixed until a homogeneous suspension is formed. The vaccine is administered in compliance with the rules of asepsis and antisepsis; sterile syringes and needles are used for administration. The injection site is treated with 70% alcohol. Symptoms of plague, infectious hepatitis, adenoviral and coronavirus infections, parainfluenza of carnivores, parvovirus enteritis and leptospirosis of dogs or other pathological signs have not been established in case of an overdose of the vaccine. Violation of the schedule (timing) of vaccination should be avoided, as this can lead to a decrease in the effectiveness of immunoprophylaxis of plague, infectious hepatitis, adenovirus and coronavirus infections, parainfluenza of carnivores, parvovirus enteritis and leptospirosis of dogs. If the next dose of vaccine is missed, immunization should be carried out as soon as possible. No interaction of the Vanguard Plus 5 L4 CV vaccine with other drugs has been identified. SIDE EFFECTS When using the vaccine in accordance with these instructions, there are usually no side effects or complications. In rare cases, anaphylaxis may occur, and symptomatic therapy is carried out. CONTRAINDICATIONS It is prohibited to vaccinate clinically ill, weakened animals and puppy bitches. Since the vaccine contains the antibiotic Gentamicin as a preservative, its use in pregnant bitches is not recommended. The use of immunosuppressants and hyperimmune serums within 1 month before and after vaccination is contraindicated. SPECIAL INSTRUCTIONS Features of the post-vaccination reaction during primary and repeated immunizations have not been established. The timing of possible use of animal products after the introduction of the Vanguard 5 L4 CV vaccine has not been established. Personal preventive measures When working with the vaccine, you should follow the general rules of personal hygiene and safety precautions provided for when working with veterinary medicinal products. All persons involved in vaccination must be provided with special clothing. At the place of work there should be a first aid kit. If the vaccine accidentally gets on the skin or mucous membranes, it must be washed off immediately with plenty of clean water. If the drug is accidentally administered to a person, the injection site must be treated with a 70% ethyl alcohol solution and seek help from a medical facility. STORAGE CONDITIONS With caution. In a dry, cool place, protected from light and out of reach of children and animals. Separated from food and feed at temperatures from 2°C to 7°C. The shelf life of the vaccine is 18 months. Freezing of the vaccine is not allowed. Vials with residual dissolved vaccine unused within 30 minutes must be disinfected by boiling for 15 minutes and then disposed of. Disposal of decontaminated vaccine does not require special precautions.
Basic schemes for using the vaccine in puppies and dogs
Vanguard is designed to ensure healthy development and prevent pathological changes in dogs caused by coronavirus and a number of dangerous diseases.
When vaccinating puppies, the following schemes are used:
- the first time at 6 weeks, the second at 9 weeks, the third at 12. The presented classic combination allows you to develop immunity after a second injection, and the third injection strengthens it. Early vaccination is recommended in tense epidemiological situations;
- vaccination at 9 weeks, and then at 11 - 12. The scheme is relevant for pets whose use was postponed for objective reasons;
- at 12 weeks, then at 16.
In adult dogs, Vanguard is used as follows:
- an adult animal that has never been vaccinated is treated twice. They give the first injection, maintain an interval of a week or two, repeat the injection;
- during revaccination, one dose of Vanguard is administered annually.
For puppies and adult dogs, take the same dosage of 1 ml, regardless of breed and weight. Compliance with the vaccination schedule guarantees 100% protection of the animal from deadly viruses.
How to administer the vaccine correctly
When carrying out vaccination, attention is paid to three aspects: preparation of the Vanguard dose, injection site, sterility of the instruments used.
Vanguard is prepared immediately before administration. For this:
- shake the bottle with the liquid component slightly;
- pump out the contents with a syringe, after wiping the surface of the lid with a disinfectant solution;
- introduce the solution into the container with the dry component;
- shake until the substances are completely dissolved.
Draw the resulting suspension into a syringe. The injection is given intramuscularly, more commonly – subcutaneously. The site where avant-garde is introduced is treated with alcohol (70%). With a subcutaneous injection, the fold is pulled back and vaccination is performed.
Be sure to adhere to the rules of asepsis and antiseptics that prevent infection from entering the body of an animal or a person, or aimed at its destruction. For this:
Always use disposable syringes, which are treated with special solutions and disposed of. When vaccinating two or more dogs, the procedure is similar. The person administering the vaccine must use rubber gloves that can prevent contact of the suspension components with the skin. It is important to have access to a first aid kit. Avoid contact of the vaccine with mucous membranes. Vanguard's solution is stored for no more than 30 minutes, then it is disinfected by boiling (10 - 15 minutes) or treated with an alkali solution (2%). Recycled.
Storage conditions and manufacturer
The vaccine should be stored in a dark and damp place at a temperature of 1 to 8 degrees Celsius. Direct sunlight or high temperature will negatively affect the activity of the vaccine. But you should not freeze the drug. After opening, use the entire dose, do not store. Use only sterile instruments when administering the vaccine, but do not wash them with chemicals. After vaccination, burn all unused contents of the bottle and used instruments. Shelf life – 1 year 6 months.
Contraindications, consequences of use
Vanguard is prohibited for use during pregnancy, because Gentamicin has an inhibitory effect on embryos.
In addition, the vaccine is not accepted:
- when feeding;
- in case of painful conditions of the animal;
- in the presence of reactions (allergic) to Vanguard components.
Weak and old dogs are not vaccinated.
The vaccine is well tolerated by puppies and adults without causing side effects. In cases where Vanguard doses were exceeded, no symptoms of infection with coronavirus or plague were recorded. Sometimes, anaphylaxis developed, the consequences of which were treated with the use of symptomatic remedies.
Expand knowledge regarding the vaccine Vanguard 5 cv vaccine!
Vanguard Plus 5 series vaccines help protect puppies from diseases that pose the greatest risk for the development of parvovirus enteritis.
- The CAV-2 component of the VANGUARD Plus 5 vaccines
cross-immunizes against CAV type 1 to help protect dogs from hepatitis without causing side effects after vaccination - including persistent kidney infections, uveitis and corneal opacity (blue eyes), which is sometimes attributed to the CAV-2 antigen. 1 containing vaccines. - The long-term effectiveness of viral antigens has been demonstrated based on retrospective serological studies using Vanguard plus 5 l4 cv.
- The consumer-friendly vaccine packaging includes tear-off labels for faster and more accurate record-keeping, a color-coded organization system and an easy-to-open safety cap.
- VANGUARD+5 is backed by our Companion Animal Immunization Support Guarantee (ISG).
Pros and cons of the Vanguard vaccine
The drug has proven effectiveness. Among its advantages are:
- good tolerance;
- creating an immune response to diseases dangerous to dogs, including plague and coronavirus;
- a simple vaccination schedule;
- convenience when preparing the suspension;
- possibility of administration at home.
Universality of dosage, suitable for all dogs regardless of age, breed, weight.
Vanguard's significant disadvantages include: the difficulty of complying with storage conditions during transportation and sales, special conditions for disposal and asepsis, and annual revaccination.